17 KiB
BEH-AXO.md
Qui comprendiamo:
- BEH-AXO: Axon
- BEH-PRE: Presynapse
- BEH-VCGG: Voltage-Controlled Gated Channels
BEH-AXO: Container
The axon does not contain specific behavior. We might add balancing of ATP within PRE later. Here we comprehend it as a “cable” transporting the AP from SOMA to Presynapse. It expands BEH-PRE which:
- can be developed by DEV-PRE
- the associations between BEH-PRE, BEH-POST e BEH-SYN is performed by the module that instantiate the Neurons and the Atrocytes, for example BEH-EXH or BEH-INH from winnertakeall.
container: BEH-AXO
expansion: BEH-PRE ( fullness: 50x, active: 0x, emptiness: 10x )
# managed_by: BEH-EXH or BEH-INH from winnertakeall
# developed_by: DEV-AXO-BEH-PRE-TUB from DEV-N
BEH-PRE: Container
The presynapse is the sending terminal of a neuron — a small bulb at the tip of an axon whose job is to release chemical signals, called neurotransmitters (NT), into the synaptic cleft, the narrow gap that separates it from the receiving neuron's postsynapse.
To do this, the presynapse maintains a stockpile of NT packed inside small membrane bubbles called vesicles. These vesicles are organised in two pools: a reserve pool (RP), which is the deep storage, and a readily-releasable pool (RRP), which is the small set of vesicles docked at the membrane and ready to fire immediately. When a spike arrives — an electrical pulse called an action potential — it briefly opens specialised calcium channels (VGCCs) in the membrane. Calcium (Ca²⁺) rushes in, and the sudden local surge of calcium triggers the docked vesicles to fuse with the membrane and pour their NT into the cleft.
But the presynapse does not just release blindly. It runs several interlocking feedback loops that continuously regulate how much it releases, how quickly it recovers, and when it should stop entirely to protect itself.
The amount of Ca²⁺ that enters is itself regulated. Three brakes — CDI, eCB, and mGluR — each reduce the effective number of open channels in their own way and on their own timescale. CDI (calcium-dependent inactivation) is a channel-level self-brake: Ca²⁺ that enters during a spike physically blocks the same channels from reopening, accumulating gradually across repeated spikes. eCB (endocannabinoids) is a retrograde signal synthesised by the receiving neuron when it is over-stimulated; it travels backward across the cleft to suppress the presynaptic channels. mGluR is a presynaptic autoreceptor that senses accumulated NT in the cleft and reduces channel conductance through a slower chemical signalling cascade.
The release of vesicles itself is regulated by two separate NT-sensing mechanisms. One acts locally at the release site in the same millisecond: high NT already in the cleft reduces how many docked vesicles fuse, trimming the current release event. The other is the mGluR pathway described above, which acts more slowly and suppresses the next spike's Ca²⁺ influx rather than the current one.
After release the vesicle stockpile must be replenished. The RRP is refilled from the RP on a timescale of seconds, at a speed that depends on recent calcium history — the synapse replenishes faster when it has been active recently. The RP itself is replenished over minutes via a chemical shuttle from the neighbouring astrocyte, a support cell that recycles the released NT back into a precursor form and ships it back to the presynapse.
The astrocyte is also the gateway to the energy supply. All of the active processes — pumping Ca²⁺ back out, docking vesicles, running the membrane pumps that restore the electrical gradient after each spike — consume ATP, the cell's energy currency. The astrocyte delivers glucose, which sets the rate of ATP replenishment. Under sustained high-frequency firing, this energy demand can outpace supply: ATP falls, the Ca²⁺ pumps slow, residual Ca²⁺ accumulates between spikes, CDI cannot recover, and the VGCCs lock shut. The synapse goes silent — not because it is broken, but because it is protecting itself from the toxic consequence of uncontrolled Ca²⁺ overload, a process known as excitotoxicity. This self-imposed silence is the central emergent behaviour we want to comprehend.
Behaviors:
— ms:
- AP fires → membrane jumps to peak, decays toward rest (Na/K-ATPase)
- ATP cost charged per AP (Na/K-ATPase recharge)
- Ca²⁺ enters via VGCCs, gated by CDI, eCB, and mGluR suppression
- Ca²⁺ buffered by calbindin / calmodulin (fast capture, slow release)
- Ca²⁺ cleared by NCX (always), PMCA and SERCA (ATP-dependent)
- ATP cost charged per unit Ca²⁺ extruded by PMCA and SERCA
- SERCA loads Ca_ER store as a side-effect of clearance
- CDI rises with Ca²⁺ — only during spike (channels open and Ca²⁺ entering)
- CDI recovers every ms — rate suppressed when Ca²⁺ is high (self-locking)
- Ca²⁺ trace (Tr_Ca) integrates every ms, including between spikes
- Vesicles release from RRP — driven by Ca²⁺ Hill sensor, suppressed by NT_cleft
- NT added to cleft
- NT_released_this_window accumulates (feeds mGluR and IP3 in seconds loop)
- NT passively diffuses out of cleft (physical, not astrocyte)
— seconds:
- Astrocyte EAATs actively clear 30% of remaining NT_cleft
- IP3 integrates NT_released_this_window (cumulative burst load)
- If IP3 exceeds threshold → astrocyte Ca²⁺ wave triggered
- mGluR autoreceptor activation updates from NT_released_this_window
- eCB retrograde signal updates from V_post history (postsynaptic input)
- RP → RRP recruitment runs (rate gated by Tr_Ca, costs ATP)
- NT_released_this_window resets to zero
— mins:
- ATP_demand (accumulated from ms loop) reduces ATP_level
- ATP_demand resets to zero
- Glucose level sets metabolic health and conversion_efficiency
- conversion_efficiency gates glutamine shuttle throughput
- Glutamine shuttle refills N_RP from astrocyte store
Semplified comprehension:
In this first comprehension, we decide to simplify:
- The VCGG are active while the AP is active, we do not comprehend the decay
- We do not comprehend the ATP
- We do not comprehend CDI, we check just for Ca2+ concentration
- We do not comprehend mGlur, we check for the concentration of NT in the cleft
- We do not comprehend Ca2+ buffering
- We do not comprehend SERCA, we comprehend Ca2+ clearing as a slow process
- We do not comprehend vesicles, we comprehend them as processes releasing NT, fast, medium and slow based on conditions
The simplification impies that:
-
Removing CDI and mGluR means Ca²⁺ concentration and NT in the cleft are now the only two conditions controlling release rate.
-
Removing ATP removes the metabolic silencing cascade entirely. The mins loop now only does one thing: replenish the NT reserve. If we want the synapse to still be able to fail under sustained firing, the mechanism would have to come from NT depletion alone (RP exhausted, nothing to replenish) rather than from pump failure and Ca²⁺ accumulation.
-
"Ca²⁺ cleared slowly" replaces PMCA, NCX, and SERCA with a single exponential decay. This means Ca²⁺ will still accumulate under high firing if the decay is slow relative to the spike rate, which preserves some of the residual-Ca²⁺ dynamic even without the full pump machinery.
Simplified Behaviors:
— ms:
- AP fires → VGCCs open, Ca²⁺ enters
- Ca²⁺ cleared slowly (single decay term, no pump detail)
- Ca²⁺ trace (Tr_Ca) integrates every ms
- NT released into cleft — rate determined by Ca²⁺ level and NT already in cleft
- fast release when Ca²⁺ high, NT_cleft low
- medium release when Ca²⁺ medium, or NT_cleft medium
- slow release when Ca²⁺ low, or NT_cleft high
- NT added to cleft
- NT_released_this_window accumulates (feeds seconds loop)
- NT passively diffuses out of cleft
— seconds:
- Astrocyte EAATs clear 30% of remaining NT_cleft
- IP3 integrates NT_released_this_window (cumulative burst load)
- If IP3 exceeds threshold → astrocyte Ca²⁺ wave triggered
- eCB retrograde signal updates from V_post history (postsynaptic input)
- eCB suppresses NT release (feeds back into ms loop release rate)
- RP → RRP recruitment runs (rate gated by Tr_Ca)
- NT_released_this_window resets to zero
— mins:
- Glucose level sets base conversion_efficiency
- If astrocyte wave was triggered → conversion_efficiency boosted temporarily
- Glutamine shuttle refills NT reserve from astrocyte store (faster if wave active, baseline if not)
- Wave boost decays back to baseline over subsequent cycles
Tubs:
- Ca2+: Calcium Ion entering the Presynapse when VCGG open that influence NT release. Normally returns to ~0 between spikes; stays elevated when pumps fail. They are key to check the concentration, release vescicles and modulation
- Rrp: Readily Releasable Pool: The Readily Releasable Pool consists of the vesicles that are "docked" and "primed" at the active zone of the synapse.
-- Location: Directly touching the presynaptic membrane.
-- Function: These are the first to be released when an action potential arrives.
-- Characteristics: This pool is very small (usually only about 0.5% to 5% of total vesicles) and can be exhausted quickly during high-frequency firing, leading to "short-term depression" of the signal. - Rp: The "Reserve Pool"
-- Location: The bulk of the vesicles held further back in the terminal, often tethered by a protein called synapsin.
-- Function: These are only mobilized during intense, prolonged stimulation once the RRP.
-- Characteristics: This makes up the vast majority of the vesicles (up to 80% or 90%). - Nt: Neuro Transmitter, released in the synapse by the vescicles
- CaTraces: sono le tracce di permanenza della concentrazione di Ca2+. Servono alla modulazione (TUN)
container: BEH-PRE
expansion: BEH-PRE-VGCC ( fullness: 10x, active: 5x, emptiness: 2x )
# tuned_by: TUN-PRE-VGCC from TUN.N
tub_local:
- Ca2+ ( fullness: 60x, active: 30x, emptiness: 0x )
# developed_by: DEV-PRE-CA2+FULL from DEV.N
- Rrp ( fullness: 30x, active: 15x, emptiness: 0x )
# developed_by: DEV-PRE-RRP-FULL from DEV.N
- Rp ( fullness: 30x, active: 15x, emptiness: 0x )
# developed_by: DEV-PRE-RRP-FULL from DEV.N
- CaTraces ( fullness: 50x, active: 0x, emptiness: 0x )
- mGluR ( fullness: 50x, active: 0x, emptiness: 0x )
tub_intricated:
- NT # SYN
- ATP # AXO?
- eCB # from POST
context_intricated:
- AP ( contained_by: BEH-SOMA )
ms: behavior
RRPConcentration: Context
Il rilascio di NT lo facciamo nel contesto di AP. Biologicamente dovrebbe avvenire solo in base alle concentrazioni, quindi anche al difuori degli AP.
context: RRPConcentration
contained_by: BEH-PRE
in_context: AP
rf: ( active: 60x )
condition: (Rrp medium)
out_context: RRPMedium
condition: (Rrp full)
out_context: RRPFull
Ca2+Concentration: Context
context: Ca2+Concentration
contained_by: BEH-PRE
in_context: AP
rf: ( active: 60x )
condition: (Ca2+ medium)
out_context: Ca2+Medium
condition: (Ca2+ full)
out_context: Ca2+Full
NTrelease: Episodes
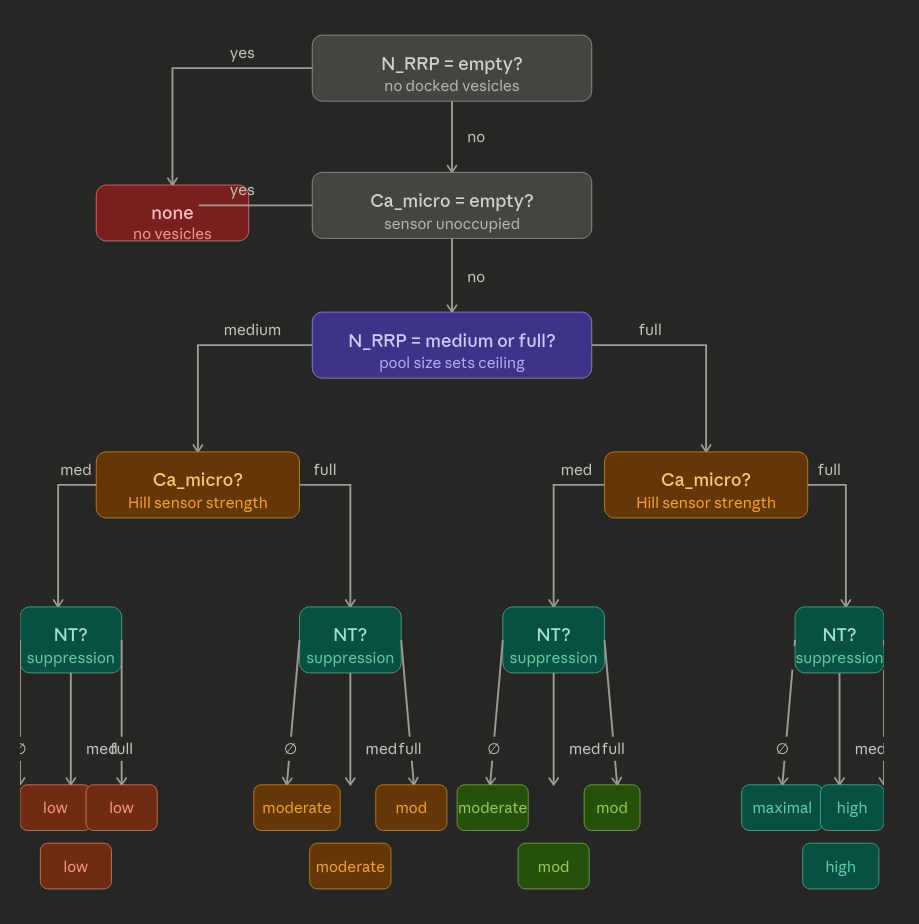
Ci sono 4 casi che dipendono da RRP, Ca2+ e NT. L'idea e' che la quantita' di RRP sia il driver principale. Gli NT liberati sono di piu' al crescere di RRP e Ca2+ e di meno al crescere di NT. Gli NT nella sinapsi fanno da moderazione alla ulteriore liberazione di NT, ma non bloccano mai totalmente. NT suppression only matters when everything else is already at maximum — which is exactly the biological purpose: it prevents runaway release during peak activity, not during moderate activity.
ATP cost of Na/K-ATPase recharge on each AP. The cost is per action potential. Here we charge it at every release of NT. This is the dominant ATP drain at high firing rates.
NTreleaseMaximum: Episode
episode: NTreleaseMaximum
contained_by: BEH-PRE
in_context: (Ca2+Full AND RRPFull)
rf: ( active: 6x ) # Maximum
hypothesis: (NT empty)
action: [Rrp decrease, Nt increase, ATP decrease]
trace: None
NTreleaseHigh: Episode
episode: NTreleaseHigh
contained_by: BEH-PRE
in_context: (Ca2+Full AND RRPFull)
rf: ( active: 6x ) # High
hypothesis: NOT (NT empty) # solo in questo caso NT modera!
action: [Rrp decrease, Nt increase, ATP decrease]
trace: None
NTreleaseMedium: Episode
episode: NTreleaseMedium
contained_by: BEH-PRE
in_context: (Ca2+Full AND RRPMedium) OR (Ca2+Medium AND RRPFull)
rf: ( active: 6x ) # Medium
hypothesis: (NT empty) OR NOT (NT empty) # In tutti i casi
action: [Rrp decrease, Nt increase, ATP decrease]
trace: None
NTreleaseLow: Episode
episode: NTreleaseLow
contained_by: BEH-PRE
in_context: (Ca2+Medium AND RRPMedium)
rf: ( active: 6x ) # Low
hypothesis: (NT empty) OR NOT (NT empty) # In tutti i casi
action: [Rrp decrease, Nt increase, ATP decrease]
trace: None
CaClearance
Qui eliminiamo Ca2+. Non comprendiamo per ora:
- PMCA: primary, ATP-dependent
- NCX: fast, NOT ATP-dependent
- SERCA: slowest, ATP-dependent
Quindi non comprendiamo anche il ristabilimento del Voltage, con altri Ioni entranti e uscenti, per ora tutto dipende da AP del SOMA.
sec: behavior
...
mGlur concentration
Limita rilascio NT: Dipende da quanti NT sono stati gia' rilasciati nella Syn
eCB concentration
Limita rilascio NT: Dipende da POST che tende a bloccare rialascio di NT se non servono
CaTrace concentration
Serve a dare la velocita' al trasporto di vesicles da RP a RRP. Ha un decadimento proprio il che dice alla Presinapsi di accellerare se da poco c'e' stata una spike, altrimenti di andare piu' piano. So after one second of silence Tr_Ca has fallen to ~37% of its peak value, after two seconds to ~14%, after three seconds to ~5%. It asymptotes toward zero but never exactly reaches it. Between spikes, Ca2+ falls toward zero as the pumps clear it.
RP->RRP shuttling
Dipende da?
min: behavior
Refill RP from Glutamine
Dipende da Glutamine messa a disposizione dall'Astrocyte
BEH-PRE-VGCC: Container
Voltage-Controlled Gated Channels: Qui per ora non gestiamo l'evoluzione della depolarizzazione. Alla scomparsa dell'AP, i VGCC smettono di funzionare.
container: BEH-PRE-VGCC
tub_intricated:
- Ca2+ ( contained_by: BEH-PRE )
context_intricated:
- AP ( contained_by: BEH-SOMA )
ms: behavior VGCC
Ca2+enter: Episodes
Here we comprehend the breaking activity on VGCC by: CDI,eCB and mGluR:
- Open — zero active brakes. mGluR alone never escapes this group because its ceiling is alpha_mGluR = 0.4, meaning even at full it only removes 40% of conductance, leaving 60% — still above the 85% threshold. So mGluR is irrelevant to the open/not-open boundary. Only CDI and eCB decide.
- Reduced/partial — exactly one meaningful brake active. Either CDI has started building (medium), or eCB has risen from sustained postsynaptic activity, but not both simultaneously. The system is aware something is happening but has not compounded yet. This is the normal operating range during moderate sustained firing.
- Suppressed — two brakes multiplying. The compounding is what defines this zone — no single variable alone produces it (except CDI approaching full). 0.5 × 0.5 = 0.25 remaining is where the synapse starts losing significant transmission efficacy. Biologically this is the pre-silence warning zone: CDI is building from residual Ca²⁺ while eCB is already engaged from postsynaptic activity.
- Closed — CDI = full is the only reliable hard rule. Because CDI can reach 1.0 and appears as (1 - CDI_factor) in the formula, it alone drives conductance to zero regardless of eCB and mGluR state. The three-brake overlap corner case (eCB=full + CDI=medium + mGluR=full) also reaches here, but in practice CDI reaching full is the primary biological mechanism.
Qui sostituiamo il CDI con Ca2+:
- CDI is calcium-dependent inactivation of VGCCs. The inactivation happens because Ca²⁺ enters through the channel and binds to a calmodulin tethered to the channel's intracellular face, physically blocking it from reopening. This is a local, channel-specific event — it requires Ca²⁺ to be flowing through that channel right now, not residual Ca²⁺ drifting in the cytosol between spikes.
- The recovery, by contrast, should run every millisecond unconditionally — CDI de-inactivation is a continuous process that proceeds whenever Ca²⁺ dissociates from calmodulin, which depends on the ambient Ca_micro level at all times.
Devo controllare che le condizioni sotto siano esaustive.
Open: Episode
episode: Open
contained_by: BEH-PRE-VGCC
in_context: AP
rf: ( active: 6x )
hypothesis: (Ca2+ empty) and (eCB empty)
action: [Ca2+ increase, ATP decrease]
trace: None
Reduced-partial: Episode
episode: Reduced-partial
contained_by: BEH-PRE-VGCC
in_context: AP
rf: ( active: 6x )
hypothesis: (Ca2+ medium) OR ((eCB medium) AND (Ca2+ empty)) OR ((eCB full) AND (Ca2+ empty) AND (mGluR empty))
action: [Ca2+ increase, ATP decrease]
trace: None
Suppressed: Episode
episode: Supressed
contained_by: BEH-PRE-VGCC
in_context: AP
rf: ( active: 6x )
hypothesis: ((Ca2+ medium) AND (eCB full) OR (eCB medium))
action: [Ca2+ increase, ATP decrease]
trace: None