18 KiB
The Biological "Cascade of Failure"
This model now demonstrates Metabolic Silencing, which is a highly consistent biological behavior:
- High firing rate → Vesicle Depletion (Fast).
- High firing rate → ATP Depletion (Slow).
- Low ATP → Pump Failure (JPMCA slows down).
- Pump Failure → Residual Calcium stays high.
- Residual Calcium → CDI stays active (The VGCCs lock shut).
- Result: The synapse stops firing to save itself from excitotoxicity.
CASCADE 1 — Vesicle Depletion appears at Max_RRP, Max_RP, p_release_base, k_rec_fast/slow, stochastic_release, map_trace_to_speed, and the recruitment block in Loop 1. The key annotation explains the asymmetry: p_release_base * Ca_micro makes each spike draw more vesicles as Ca_micro rises early in a burst — a positive feedback that accelerates the collapse before recruitment can respond.
CASCADE 2 — ATP Depletion is anchored at Glucose_level (the root input) and at compute_astrocyte_metabolic_health in Loop 3, where it explains that ATP_level is the bridge variable that carries minute-scale metabolic state into the millisecond Ca²⁺ world.
CASCADE 3 — Pump Failure is annotated at k_PMCA, k_NCX, k_SERCA, ATP_half, and compute_pump_atp_factor. The NCX comment explicitly notes its role as a floor-not-rescue — it keeps clearing during failure and enables the auto-reset, but cannot prevent accumulation alone.
CASCADE 4 — Residual Ca²⁺ appears at B_total, tau_buffer_rebind, the capture_fraction block, and the buffer recharge lines. The buffer saturation note explains the two-phase dynamic: buffer is protective early but becomes invisible once B_free → 0.
CASCADE 5 — CDI Lock-out is annotated at k_CDI_rise, Ca_micro_saturation, k_CDI_rec, and both the rise and recovery lines in Loop 1. The recovery comment spells out the self-locking logic explicitly as a chain.
CASCADE 6 — Silence sits at effective_conductance with a timing note showing that mGluR fires first, eCB second, and CDI last but irreversibly. The eCB and mGluR annotations in Loop 2 explain their roles as early partial brakes versus the terminal lock.
5. Model Summary Checklist
- [x] Timing: Spans 0.1 ms (AP) to 300,000 ms (Metabolism).
- [x] Conservation: Vesicles and Neurotransmitters are conserved through the Gln→RP→RRP→Cleft→Astro loop.
- [x] Stability: CDI and mGluR/eCB provide three layers of negative feedback to prevent runaway excitation.
- [x] Resource Constraints: ATP and Pool guards prevent physically impossible negative values or infinite accumulation.
This pseudocode serves as a comprehensive blueprint for a biologically consistent tripartite synapse. It is structured into three nested temporal loops to handle the transition from microseconds to minutes.
Variable Reference Table
| Variable | Definition | Scale | Role |
|---|---|---|---|
| Ca_micro | Free calcium in the active zone | 0.1 ms | Triggers release and CDI |
| B_free | Available buffer sites (Calbindin) | 0.1 ms | Immediate calcium "sponge" |
| N_RRP | Readily Releasable Pool | 1 ms | Immediate vesicle supply |
| N_RP | Reserve Pool | 100 ms | Long-term vesicle warehouse |
| Tr_Ca | Calcium Trace | 10 ms | Memory of recent activity |
| CDI_fac | Inactivation Factor | 50 ms | Internal negative feedback |
| mGluR_pre | Autoreceptor activation | 500 ms | Cleft-sensing inhibition |
| ATP_level | Metabolic energy state | 1 min | Gates calcium clearance |
The Multi-Scale Engine
# --- SIMULATION CONFIGURATION ---
# dt = 0.1 ms (Fine-grained step)
# Total_Time = 300,000 ms (5 minutes)
while t < Total_Time:
# ============================================================
# 1. FINE-GRAINED NEURAL LOOP (Every 0.1ms)
# Handles: Electrical spikes, Buffering, and Influx
# ============================================================
# --- ACTION POTENTIAL & WAVEFORM ---
if is_AP_active(t):
# Layered Inhibition logic:
# CDI (Internal), eCB (Retrograde/Post), mGluR (Autoreceptor/Cleft)
total_inhibition = (1 - CDI_fac) * (1 - eCB_level) * (1 - mGluR_pre * alpha_mGluR)
# Calculate Influx via VGCC
# V_pre_pulse(t) accounts for the finite duration of the spike window
raw_influx = N_VGCC * total_inhibition * V_pre_pulse(t)
# --- FAST BUFFERING BEHAVIOR ---
# Immediate capture of influx by buffer proteins (e.g., Calbindin)
captured = raw_influx * (B_free / B_total)
B_free = max(0, B_free - captured)
Ca_bound += captured
# Resulting free Calcium that actually reaches the sensors
Ca_micro += (raw_influx - captured)
# --- STOCHASTIC RELEASE ---
if N_RRP > 0:
# Release probability is a function of Ca_micro
p_release = compute_stochastic_p(Ca_micro, N_RRP)
if random_uniform(0, 1) < p_release:
N_RRP -= 1 # Deplete one vesicle
Glu_cleft += 1 # Release NT into cleft
CDI_fac += k_CDI_rise # Increment inactivation per release
# --- CONTINUOUS CALCIUM CLEARANCE ---
# NCX (Sodium-Calcium Exchanger) - Fast, gradient driven
# PMCA (Plasma Membrane Ca-ATPase) - Slow, ATP dependent
atp_efficiency = ATP_level**2 / (ATP_level**2 + 0.3**2)
cleared = (k_NCX * Ca_micro) + (k_PMCA * Ca_micro * atp_efficiency)
Ca_micro = max(0.0, Ca_micro - cleared)
# --- RECOVERY MECHANISMS ---
# CDI Recovery: Decay of inactivation as Ca_micro falls
CDI_fac = max(0.0, CDI_fac - (dt / tau_CDI_rec))
# Buffer Recovery: Re-release of bound ions into microdomain
re_release = Ca_bound * (dt / tau_buf_release)
Ca_bound -= re_release
Ca_micro += re_release
B_free = B_total - Ca_bound
# ============================================================
# 2. MID-GRAINED INTEGRATION (Every 10ms - 100ms)
# Handles: Recruitment Traces and Autoreceptor Feedback
# ============================================================
if t % 10 == 0:
# TRACE INTEGRATOR: The memory of recent spikes
Tr_Ca = update_leaky_integrator(Tr_Ca, Ca_micro, tau_trace)
# RECRUITMENT LOGIC (RP -> RRP)
# Recruitment speed (k_rec) scales non-linearly with Tr_Ca
k_rec = compute_k_rec(Tr_Ca)
# Apply HARD CAPS and GUARDS:
# 1. Cannot take more than what is in RP
# 2. Cannot exceed the ceiling of RRP
refill_qty = k_rec * N_RP * (Max_RRP - N_RRP)
refill_qty = max(0, min(refill_qty, N_RP))
N_RRP += refill_qty
N_RP -= refill_qty
# AUTORECEPTOR FEEDBACK: Presynapse sensing its own NT
mGluR_pre += (Glu_cleft / (Glu_cleft + Km) - mGluR_pre) * (10 / tau_mGluR)
# ============================================================
# 3. COARSE-GRAINED METABOLIC LOOP (Every 1s - 1min)
# Handles: Astrocyte support, eCB Brake, and Sustainability
# ============================================================
if t % 1000 == 0:
# ASTROCYTE GLUTAMATE CLEARANCE
# Astrocytes clean the cleft; NT is recycled into the Glutamine pool
cleared_glu = Glu_cleft * EAAT_clearance_rate
Glu_cleft -= cleared_glu
Gln_pool += cleared_glu
# RETROGRADE BRAKE (eCB from Postsynapse)
# Postsynapse synthesizes eCB based on its own V_post activity
eCB_level = update_retrograde_brake(V_post_history)
# METABOLIC REPLENISHMENT
# Astrocyte health determines ATP; Glutamine refills the Reserve Pool
ATP_level = compute_atp_from_astro_health(Gln_pool, Metabolic_State)
# Long-term Refill of the Reserve Pool (The Warehouse)
N_RP = min(N_RP + (Gln_pool * metabolic_shuttle_rate), Max_RP)
Gln_pool *= 0.9 # Account for metabolic overhead/loss
t += dt # Increment simulation time
3. Biological Consistency Summary
- Metabolic Coupling: The
atp_efficiencyvariable creates a physical link between the 5-minute astrocyte clock and the 0.1ms calcium clock. If the astrocyte is exhausted, the pumps fail, and theCDI_faclocks the synapse into silence. - Double-Lock Inhibition: You have two distinct brakes. The
mGluR_preis "homosynaptic" (local self-check), while theeCB_levelis "heterosynaptic" (feedback from the receiver). This prevents runaway excitation and excitotoxicity. - Conservation of Mass: Neurotransmitters follow a closed loop:
Gln_pool->N_RP->N_RRP->Glu_cleft->Astrocyte->Gln_pool. - Buffer Buffer: The
B_freemechanism prevents numerical instability during high-frequency bursts by absorbing excess calcium influx instantly.
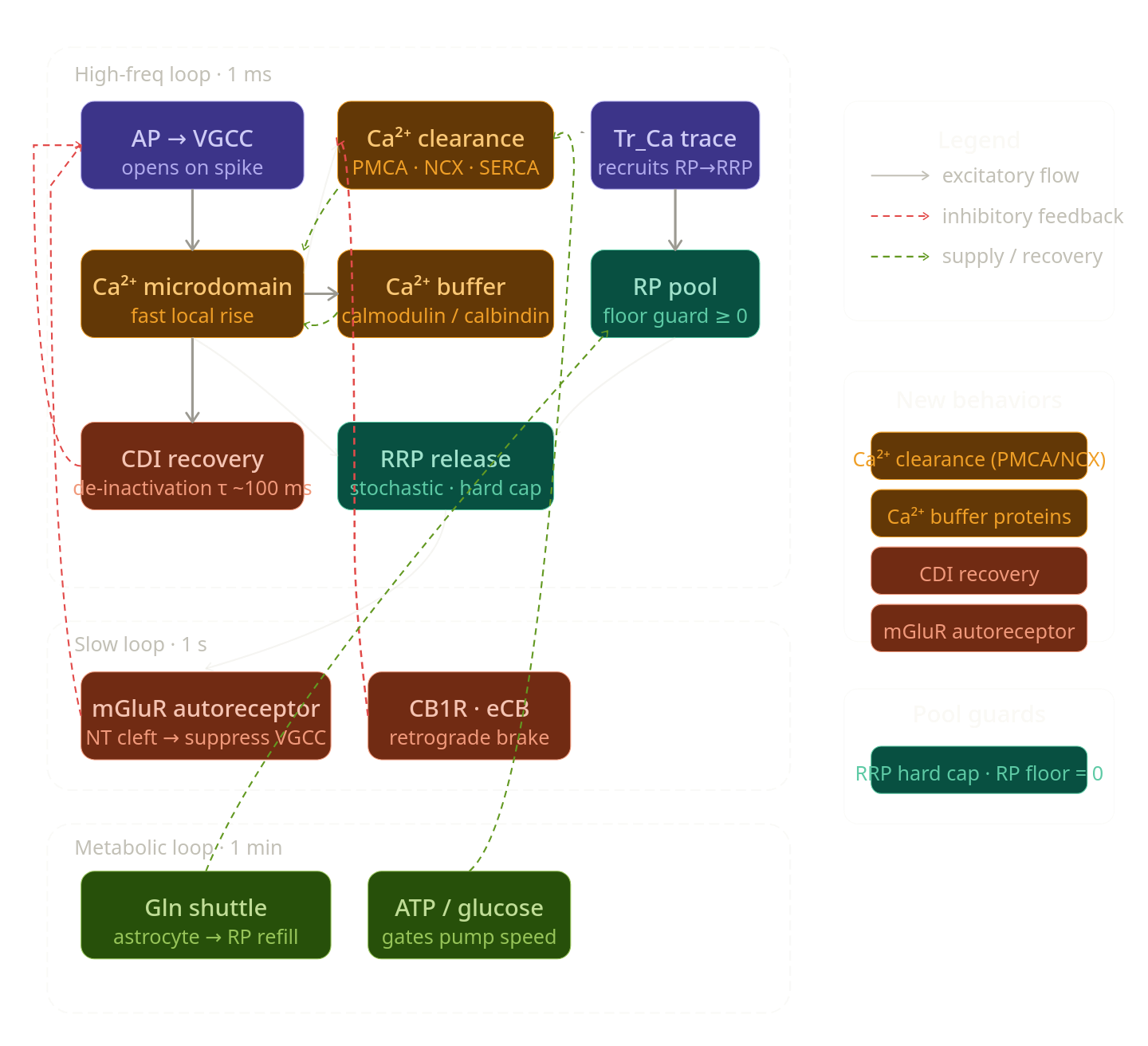
The missing presynaptic behaviors are:
- Ca²⁺ clearance (PMCA, NCX pumps) — without this, Ca²⁺ accumulates indefinitely
- Calcium buffer proteins (calbindin, calmodulin) — fast buffering before pumps clear
- VGCC inactivation recovery — CDI is mentioned but the recovery (de-inactivation) is missing
- RRP hard cap — the refill logic can overshoot without a ceiling
- Reserve Pool (RP) depletion guard — refill can go negative
- Presynaptic autoreceptors (mGluR/CB1R feedback closing the loop from NT in cleft → presynaptic suppression) — separate from eCB which comes from postsynapse
- Spike refractory / AP waveform duration — the effective window for Ca²⁺ entry is finite
Let me build an annotated code diagram and then write the enhanced model.Here is a full analysis of every missing loop, followed by the updated code for each section.
Missing behaviors and the reasoning behind each
Why Ca²⁺ clearance is the most critical gap. Ca_micro currently has no exit route — it only grows. Without PMCA pumps, NCX exchangers, and SERCA (ER uptake), every spike leaves residual Ca²⁺ that accumulates across the simulation and eventually locks the synapse in a permanently over-activated (or CDI-locked) state. The clearance mechanisms also run at different speeds: NCX is fast (tens of ms), PMCA is slower but higher-capacity, SERCA is slowest and stores calcium for later use as an internal buffer.
Why Ca²⁺ buffer proteins must precede clearance. Calbindin and calmodulin bind free Ca²⁺ within microseconds and act as a fast, temporary "sponge". They blunt the initial Ca_micro peak, protecting against excess CDI. They also slowly release Ca²⁺ back into the cytosol, which feeds the trace integrator more smoothly. Without buffers, the microdomain pulse is unrealistically sharp.
Why CDI recovery closes a loop without itself. The model already writes CDI_factor but never resets it. A VGCC that inactivated on spike N stays inactivated on spike N+1. CDI recovery is simply a decay back toward zero, with a time constant of ~100 ms, driven by Ca²⁺ falling (i.e., it depends on clearance — another reason clearance comes first).
Why mGluR autoreceptors are needed. The eCB pathway is a retrograde signal synthesized by the postsynapse. But the presynapse also has its own direct cleft-sensing system: presynaptic mGluR2/3 receptors bind glutamate in the cleft and suppress VGCC conductance and cAMP. This is a homosynaptic feedback loop that is entirely local to the presynapse and missing from the current model.
Why pool guards matter. The refill_amount calculation can produce N_RP < 0 if current_recruitment_rate * N_RP > N_RP. The N_RRP overshoot is subtler but also real: if two slow-loop updates stack before the fast loop consumes RRP, you can exceed Max_RRP.
Updated code, section by section
Loop 1A — Ca²⁺ dynamics (replaces the current Ca_micro += block)
# --- PRESYNAPTIC Ca2+ DYNAMICS ---
if V_pre == 1: # AP arrives
effective_conductance = N_VGCC * (1 - eCB_level) * (1 - CDI_factor)
raw_influx = compute_flux(effective_conductance, V_pre_voltage)
# ADDED: Buffer proteins capture a fraction of influx immediately.
# Buffering capacity (B_free) depletes on capture, recovers slowly.
# VARIABLE: B_free – free buffer sites (calbindin/calmodulin)
# TIMING: rebinds saturated buffer in ~200 ms
captured = raw_influx * (B_free / B_total) # fraction caught
B_free = max(0, B_free - captured) # buffer saturates
Ca_micro += (raw_influx - captured) # only free Ca2+ counts
# --- ADDED: Ca2+ CLEARANCE (runs every ms, not just on spike) ---
# Three parallel mechanisms, each with its own rate constant:
# k_PMCA ~0.03 /ms (plasma membrane Ca-ATPase, ATP-dependent)
# k_NCX ~0.10 /ms (sodium-calcium exchanger, voltage-sensitive, fast)
# k_SERCA ~0.01 /ms (ER pump, slowest, fills internal Ca2+ store)
# ADDED: ATP gates pump speed — shared with metabolic loop below
pump_scale = compute_pump_atp_factor(ATP_level) # 0→1
cleared_PMCA = k_PMCA * Ca_micro * pump_scale
cleared_NCX = k_NCX * Ca_micro # NCX is not ATP-dependent
cleared_SERCA = k_SERCA * Ca_micro * pump_scale
Ca_micro -= (cleared_PMCA + cleared_NCX + cleared_SERCA)
Ca_micro = max(0.0, Ca_micro) # hard floor
# ADDED: SERCA fills an internal ER store (Ca_ER).
# This store can be released later (e.g. mGluR activation triggers IP3→ER release).
# For now it is simply a sink; ER-release can be wired later.
Ca_ER += cleared_SERCA
# ADDED: Buffer recharge — captured Ca2+ slowly re-releases back to cytosol,
# and free buffer sites recover as Ca2+ is extruded.
# TIMING: tau_buffer_rebind ~200 ms
Ca_micro += Ca_buffer_bound * dt / tau_buffer_rebind
Ca_buffer_bound *= (1 - dt / tau_buffer_rebind)
B_free = B_total - Ca_buffer_bound # bookkeeping
Loop 1B — CDI recovery (adds a reset that was missing)
# --- CDI INACTIVATION + RECOVERY ---
# EXISTING: CDI_factor rises with Ca_micro on each spike.
CDI_factor += map_calcium_to_inactivation(Ca_micro)
# ADDED: CDI_factor decays back to zero as Ca2+ is cleared.
# VARIABLE: tau_CDI_recovery ~100 ms
# LOGIC: Recovery tracks Ca_micro level — low Ca2+ → fast de-inactivation.
CDI_recovery_rate = k_CDI_rec * (1 - Ca_micro / Ca_micro_saturation)
CDI_factor = max(0.0, CDI_factor - CDI_recovery_rate * dt)
Loop 1C — Pool arithmetic with guards
# --- RRP RELEASE (with hard cap) ---
if N_RRP > 0:
released_NT = stochastic_release(N_RRP, Ca_micro)
N_RRP = max(0, N_RRP - released_NT)
add_NT_to_cleft(released_NT)
# --- RP → RRP RECRUITMENT (with floor and ceiling guards) ---
current_recruitment_rate = map_trace_to_speed(Tr_Ca)
refill_amount = current_recruitment_rate * N_RP * (Max_RRP - N_RRP)
refill_amount = max(0.0, refill_amount) # ADDED: never negative
refill_amount = min(refill_amount, N_RP) # ADDED: can't take more than RP holds
N_RRP = min(N_RRP + refill_amount, Max_RRP) # ADDED: hard ceiling
N_RP = max(0.0, N_RP - refill_amount) # ADDED: hard floor
Loop 2 — mGluR autoreceptor (new, 1 s loop)
# --- ADDED: HOMOSYNAPTIC AUTORECEPTOR FEEDBACK ---
# VARIABLE: mGluR_activation – presynaptic mGluR2/3 occupancy (0→1)
# TIMING: rises in ~500 ms when NT_cleft is high, decays in ~2 s
# LOGIC: Directly reduces VGCC conductance AND suppresses cAMP
# (cAMP pathway gates RRP docking speed — can be added later).
# This loop is distinct from eCB: it is local, homosynaptic, and faster.
mGluR_activation += (NT_cleft / (NT_cleft + Km_mGluR) - mGluR_activation) * (dt_slow / tau_mGluR)
# The suppression factor enters the high-freq loop at Line 1A:
# effective_conductance = N_VGCC * (1 - eCB_level) * (1 - CDI_factor) * (1 - mGluR_activation * alpha_mGluR)
#
# alpha_mGluR: max fractional suppression (~0.4 for mGluR2/3 at physiological concentrations)
Loop 3 — ATP dependency on pumps (links metabolic health to Ca²⁺ clearance)
# --- ADDED: ATP GATES CA2+ PUMP SPEED ---
# VARIABLE: ATP_level – normalized 0→1 (computed from astrocyte metabolic health)
# LOGIC: PMCA and SERCA are ATP-dependent.
# When ATP_level drops, Ca2+ clearance slows → Ca_micro stays elevated →
# CDI rises → effective VGCC conductance collapses → synapse silences.
# This is the realistic metabolic-silence cascade.
def compute_pump_atp_factor(ATP_level):
# Hill function: half-maximal at ATP_half = 0.3
return ATP_level**2 / (ATP_level**2 + ATP_half**2)
# The slowest metabolic loop already writes ATP_level via compute_astrocyte_metabolic_health().
# No further wiring needed — pump_scale above picks it up automatically.
The closed loop, stated plainly
Every Ca²⁺ that enters now has exactly one exit: PMCA, NCX, or SERCA. Buffers slow the peak. CDI rises with Ca²⁺ and falls as Ca²⁺ falls — it can no longer lock permanently. The cleft-sensing mGluR autoreceptor gives the presynapse its own, faster brake independent of the postsynapse. Pool arithmetic is bounded on both ends. And ATP depletion now cascades naturally: less ATP → slower pumps → higher residual Ca²⁺ → more CDI → fewer effective VGCCs → silence — which is precisely the metabolic-fatigue endpoint the deep loop was trying to express but couldn't reach without the pump link.